 
The half-life for a zero-order reaction isīy plotting a graph between t 1/2 on the y-axis and o on the x-axis if a straight line is obtained the reaction is zero order. The order of reaction can also be determined by using half-life.Different drugs have different half-lives but all follow the definition of the half-life: after one-half life has passed, 50% of the initial amount of drug is removed from the body.Using the concept of half-life excretion rates and steady-state concentrations for any specific drug can be determined.The concept of a half-life has also been applied for pesticides in plants describing dissipation from plants.Half-life is important in nuclear physics to describe how long unstable atoms exist and how quickly they undergo radioactive decay.The half-life for second-order reaction with different concentration of reactants cannot be determined because, so the times takes to consume one half of will not be equal to time takes to consume one half of. If the half-life is short means reactants will be consumed in a short period of time. This equation shows that half-life depends on bothįrom the equation, it is concluded that if the concentration of reactants is less, its half-life will be more, which means reactants exist for a longer period of time. K 2=rate constant for second-order reaction and o=initial concentration of reactant. The integrated rate law for 2nd order reaction with the same concentration isįor half-, life second-order put t=t 1/2 and t= o/2 The half-life for a second-order reaction Half-life is a time that is required for reactants to consume one-half of their initial concentration.įor example, if the concentration of the reactant is 12×10 -3moldm -3 at the start of the reaction, then during the first half-life concentration of reactant drops to 6×10 -3moldm -3, during second half-life concentration of reactant drops to 3×10 -3moldm -3, during third half-life concentration of reactant drop to 1.5×10 -3moldm -3. From the intercept we can calculate the value of c.Įq(2), eq(7), and eq(8) can be plotted in various ways to show different relations. From the slope of the graph we can calculate the value of the rate constant.ģ. They give information about the relation between the second-order order concentration of reactants or products with time.Ģ. 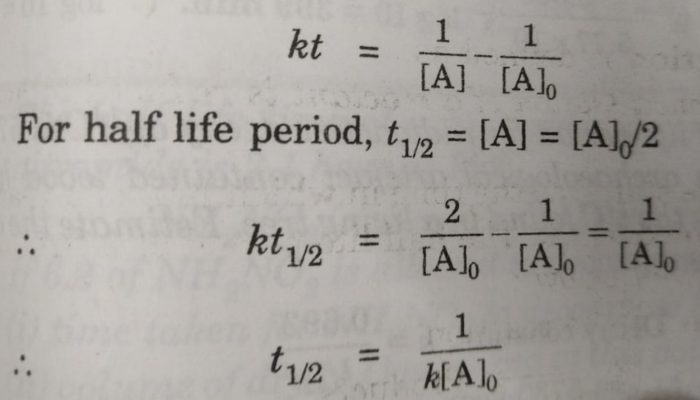
This is called an integrated form of a second-order reaction with different concentrations. When the concentration of reactants are differentĪt time t=0sec concentration of reactants areĪpply boundary conditions when t=0 x=0 in eq(6) This is called an integrated form of second-order reaction. 
This is a differential equation for second-order reactions. When the concentration of reactants is the sameĬonsider two reactants that combine to give products.Īt time t=0 concentration of reactants are oo The half-life for a second-order reaction.2nd order reaction with different initial concentrations.2nd order reaction with same initial concentrations.There are two cases for second-order reactions. Half-life is constant over the lifetime of an exponentially decaying quantity, and it is a characteristic unit for the exponential decay equation. Rutherford applied the principle of a radioactive element’s half-life to studies of age determination of rocks by measuring the decay period of radium to lead-206. The original term, half-life period, dating to Ernest Rutherford’s discovery of principle in 1907, was shortened to half-life in the early 1950s. Formation of urea from NH 4 + and CNO –.The reaction between CH 3I and C 2H 5ONa gives ether. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
